Valence Electron
- A valence electron is for holding two or more atoms together in a chemical bond.
Types of Bonds
Ionic Bonds
Na and O Na+1 O-2 Na+1 Metallic Bonds
|
Covalent Bonds
Types of Covalent Bonds:Single Bonds: 2 electrons
Double Bonds: 4 electrons Triple Bonds: 6 electrons
|

The number on the top of the periodic table is telling when the element is either losing or gaining their ions. Each number on top is for every column and is use for only that column.

<---Example of the formula for the ion is formed when the elements loses its valence electron.

- This is an examples how to solve ionic bonding.
- First, Be+2 and P-3 cancel out leaving P-1.
- Second, add another Be+2 to cancel P-1, which there is still Be+1 left.
- Then add another P-3 to cancel out Be+1, which there is still P-2 left.
- This is the last step because now Be+2 cancel out P-2 and know it is balance.
- Then write it into a formula unit.
- That is how you solve for a ionic bonding.
Formula Unit
- Formula Unit are the simplest ratio of elements in an ionic compound.
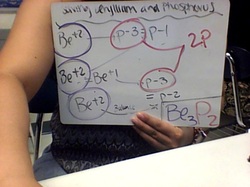
- How to count the formula unit:
<---- Example
Lewis Dot Structure
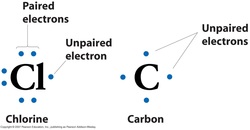
A diagram that shows the bonding between atoms of a molecule and lone pairs of electrons.
https://classes.lt.unt.edu/Fall_2010/CECS_5030_026/mrp0113/imageswebproject/Lewis%20Dot%20symbol%20examples.jpg
https://classes.lt.unt.edu/Fall_2010/CECS_5030_026/mrp0113/imageswebproject/Lewis%20Dot%20symbol%20examples.jpg
Molecular Compounds
- Compounds that are bonded covalently ( like water and carbon dioxide) are molecular compounds.
- Molecular compounds tends to be gases or liquids at room temperature.
- Molecular compounds tend to have relatively lower boiling points than ionic compounds.
Work Cited Page
- I drew the pictures and use my own examples to show.
- http://en.wikipedia.org/wiki/Lewis_structure
