Unit 4 Vocabulary
By DynaBlast (Created with Inkscape) [CC-BY-SA-2.5 (http://creativecommons.org/licenses/by-sa/2.5)], via Wikimedia Commons
Anion - A negatively charged ion.
Bond - That which holds together atoms in molecules and ions in lattices.
Cation - A positively charged ion.
Coulomb's Law - A mathematical formula whose consequence is that negatively and positively charged particles attract each other and similarly charged species repel each other.
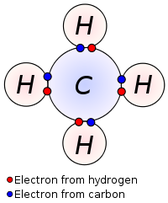
Covalent Bond - A bond that results from a sharing of electrons between nuclei.
Ion - A charged species created by the gain or loss of an electron from an atom or neutral molecule.
Ionic Bond - A bond that results from electrostatic attraction between oppositely charged ions. The cation is positively charged, while the anion is negatively charged.
Lattice - A regularly repeating three-dimensional array of atoms, molecules, or ions.
Molecular Orbital - A combination of atomic orbitals in molecular orbital theory that provides an orbital description of a molecule analogous to the atomic orbital description of atoms.
Molecular Orbital Theory - A description of bonding that combines atomic orbitals from each bondedatom to produce a set of molecular orbitals.
Molecule - A chemical species containing a covalent bond.
Valence Shell Electron Pair Repulsion Theory - A theory used to predict bonding geometries that states that electron pairs will be distributed about the central atom to minimize electron pair repulsions.
Bond - That which holds together atoms in molecules and ions in lattices.
Cation - A positively charged ion.
Coulomb's Law - A mathematical formula whose consequence is that negatively and positively charged particles attract each other and similarly charged species repel each other.
Covalent Bond - A bond that results from a sharing of electrons between nuclei.
Ion - A charged species created by the gain or loss of an electron from an atom or neutral molecule.
Ionic Bond - A bond that results from electrostatic attraction between oppositely charged ions. The cation is positively charged, while the anion is negatively charged.
Lattice - A regularly repeating three-dimensional array of atoms, molecules, or ions.
Molecular Orbital - A combination of atomic orbitals in molecular orbital theory that provides an orbital description of a molecule analogous to the atomic orbital description of atoms.
Molecular Orbital Theory - A description of bonding that combines atomic orbitals from each bondedatom to produce a set of molecular orbitals.
Molecule - A chemical species containing a covalent bond.
Valence Shell Electron Pair Repulsion Theory - A theory used to predict bonding geometries that states that electron pairs will be distributed about the central atom to minimize electron pair repulsions.
SparkNotes Editors. “SparkNote on Introduction to Chemical Bonding.” SparkNotes.com. SparkNotes LLC. n.d.. Web. 30 Apr. 2014.